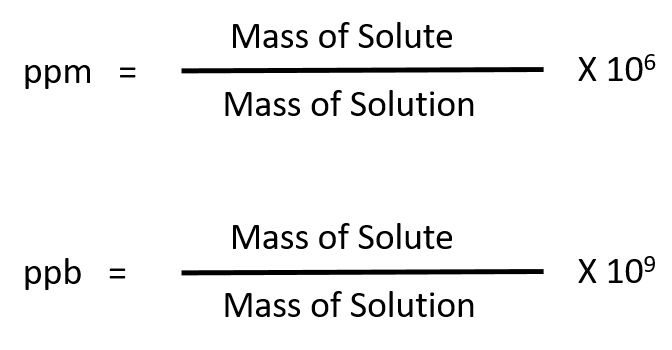
This is a great question: what are total dissolved solids that are in your drinking water or water? Even if you run a total dissolved solids test, you still do not know what the actual dissolved solids are and having total dissolved solids in your water is not good or bad. To prepare stock standard solution in lab you should first make sure that you understand the concentration unit principles and some mathematical rules which will help to find exact answer for reporting.Get Informed | Total Dissolved Solids (TDS) Elevated Total Dissolved Solids can result in your water having a bitter or salty taste result in incrustations, films, or precipitates on fixtures corrosion of fixtures, and reduced efficiency of water filter and equipment What is Total Dissolved Solids (TDS)? All stock standards shall be checked before use with another standard that has been prepared separately from different source. Example of stock standard solutions for like Sodium (Na) solution with a concentration of 1000 mg Na /L (ppm) used as stock solution for Sodium analysis by Inductively Coupled Plasma (ICP), Flame Photometry, Ion Chromatography (IC)…etc, that can be stored at 4 0C for a minimum of 6 months. Stock standard solution is defined as a solution with high concentration of stable analyte(s) that can be stored at specific conditions in laboratory for long time and used as a standard reference material for analysis of the target analyte(s) in the daily use. The accuracy in the preparation of stock standard reflects accuracy of the results. It is include some basic principles and guide to work in lab for preparing stock standard solution.Īnalysis in any laboratory mainly based on reference materials like Stock standard solution. Chapter three include practical laboratory guide. Because some chemists still can’t distinguish between them. Calculations for each type are slightly same but we make it in different sections to simplify the idea of stock standards preparation. Chapter two consider types of stock standards depending on the formula types of analyte, because of different applications in lab and different methods used in analysis, some by ICP for metals, titration or Ion Chromatography for anions and cations and Formula standards for preparation of reagents. Including a lot of examples in each chapter is to help readers to fulfill understand principles in the preparation of standard solution. It also considers view examples in each section and general example at the end of chapter. Chapter one is an introduction explain basic principles of calculations for concentration units and conversion between them. It will start from basic calculations up to practical preparation procedure in lab. It will help student, teachers, chemist, scientist and engineers … etc who’s working in chemistry field to fully understand the preparation of stock standard solution. Who Should Read it? This help book is written to be readable by anyone who has idea in basic principles of general chemistry. This simple book is a practical training for chemist, technicians or scientist for solutions calculations.Īll Copyrights reserved to Chemiasoft ©2004 – 2011, Page 6 of 61 We found some chemists are making some mistakes in calculation. We are trying our best starting from small principles in chemistry. 61įorward We are looking to make chemistry simple, easy for student, teacher, chemist, analyst and scientist.

57Ĭhecklist for preparing solutions of known concentration. 51Īll Copyrights reserved to Chemiasoft ©2004 – 2011, Page 4 of 61īasic Equipment and Instrumentation. 47 Chapter 3: Laboratory Practical Guide. 35 Chapter 2: Types of Stock Standard Solutions. 22Ĭonverting Between Concentration Units. Weight, Volume, and Weight-to-Volume Ratios. Multiplication and Division of Significant Figures. 8 1.3.1Īddition and Subtraction of Significant Figures. Guide To Preparation Of Stock Standard SolutionsĪll Copyrights reserved to Chemiasoft ©2004 – 2011, Page 2 of 61Īll Copyrights reserved to Chemiasoft ©2004 – 2011, Guide to Preparation of Stock Standard Solutions, First Edition Guide to Preparation of Stock Standard Solutions, First Edition
Guide To Preparation of Stock Standard Solutions First Edition May 2011
